 It dissociates in an aqueous solution and produces hydronium H+, PO 4– ions. Question : What are the reaction of Phosphoric acid and Calcium hydroxide?Īnswer: The reaction of Phosphoric acid and Calcium hydroxide, Ammonia, copper hydroxide, aluminum hydroxide, etc. Weak bases: Partially dissociates in an aqueous solution to form hydroxide ion. Benzoic acid, formic acid, oxalic acid etc. 
Weak acids: Partially dissociates in an aqueous solution to form protons. Question : What are the examples of weak acids and weak bases?Īnswer: The examples of weak acids and weak bases are, dissociates completely in an aqueous solution to form hydroxide ion. Sodium hydroxide, potassium hydroxide, strontium hydroxide, lithium hydroxide, etc. Question : What are the examples of strong bases?Īnswer : The examples of strong bases are, example, sulphuric acid, nitric acid, hydrochloric acid, phosphoric acid etc. The chemical species completely dissociates into an aqueous solution and produces hydronium ion or proton H+. H+ and OH- linked to form water as well as strontium ion and chloride ion linked to form strontium chloride.Īlso Read On: Is HBr Ionic or Covalent : Why? How, Characteristics and Detailed Facts Frequently Asked Questions: Question : What is a strong acid? Strontium hydroxide dissociates to form Sr+ and OH. Those chemicals are very dangerous.Being a strong acid, Hydrochloric acid dissociates to form H+ and Cl. Bases with pH values near 14 include drain cleaner and sodium hydroxide (NaOH). There are also very strong acids with pH values below 1, such as battery acid. If you ever go into a chemistry lab, you could find solutions with a pH of 1 and others with a pH of 14. When you start looking at the pH of chemicals, the numbers can go to the extremes. They are either a little below or a little above that mark. Most of the liquids you find every day have a pH near 7. Distilled water is 7 (right in the middle). The scale measures values from 0 all the way up to 14. Although there may be many types of ions in a solution, pH focuses on concentrations of hydrogen ions (H +) and hydroxide ions (OH -). Scientists use something called the pH scale to measure how acidic or basic a liquid is. Chemists use the word " dissociated" to describe the breakup of a compound. That hydrogen ion is the reason it is called an acid. (Okay, that gives away the answer.) If you look at its atoms when it's in water, you will see the molecule CH 3COOH split into CH 3COO - and H +. For example, vinegar is also called acetic acid. Those two special ions determine whether you are looking at an acid or a base. When a hydroxide ion is released, the solution becomes basic. When a hydrogen ion is released, the solution becomes acidic. 
At other times, you find the release of an OH - (hydroxide) ion. He saw that when you put molecules into water, sometimes they break down and release an H + ( hydrogen) ion. So what makes an acid or a base? A chemist named Svante Arrhenius came up with a way to define acids and bases in 1887. But what about baking soda? When you put that in water, it creates a basic solution. In fruits there is something called citric acid. In your body there are small compounds called amino acids. Those special ions in solution make something acidic or basic. Most water you drink from the tap has other ions in it. Most of the time, the positive and negative ions in distilled water are in equal amounts and cancel each other out. 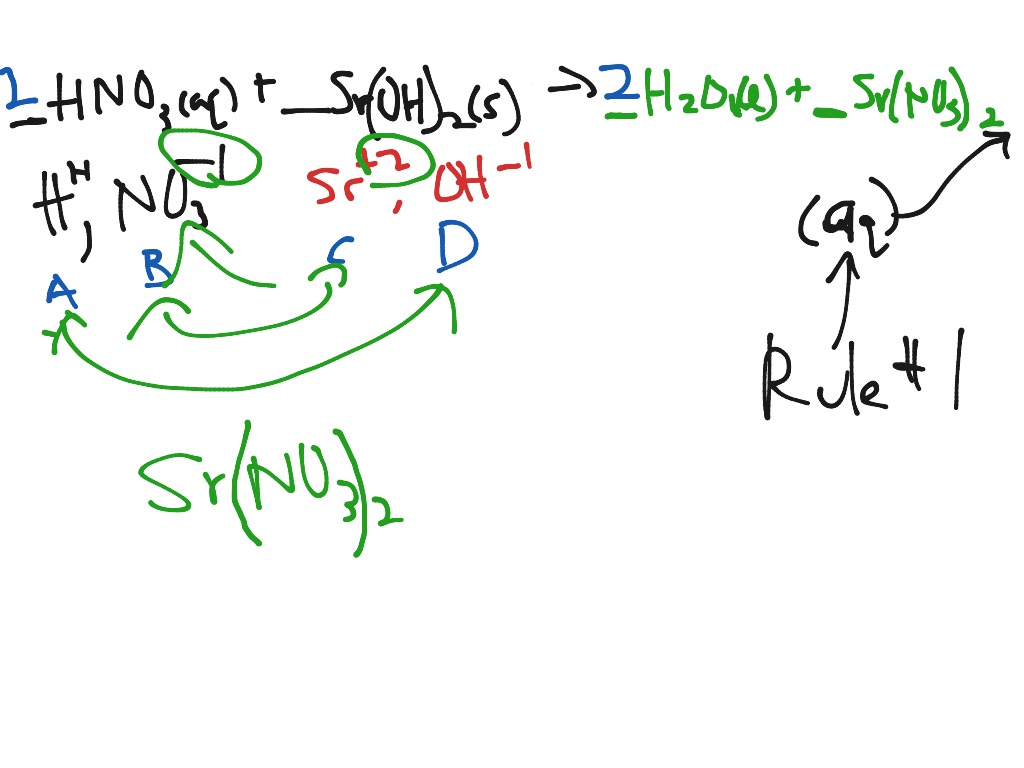
See how the hydrogen ion was transferred? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
